This process has the potential to produce devices that may become powerful tools for new challenging applications such as early cancer detection. As the pathway for the development and introduction of novel technologies to the clinical diagnostics market requires hundreds of millions of dollars and decades of work, the improvement and further development of already established LFA technologies is a favourable alternative. LFAs are very good candidates as they are cheap to produce, easy to use and, importantly, widely accepted by users and regulatory authorities. Such assays (potentially a single LFA) should be easy to perform without the use of laboratory investigation, or individuals trained in chemical analysis. In recent years there has been an increasing demand for point-of-care multiple diagnostic assays with multiple test lines allowing the rapid and simultaneous detection of multiple analytes present in samples. In these areas of utilization, rapid tests are used to screen for animal diseases, pathogens, chemicals, toxins and water pollutants, among others. Further industries in which LFA-based tests are employed include veterinary medicine, quality control, product safety in food production, and environmental health and safety. A variety of biological samples can be tested using LFAs, including urine, saliva, sweat, serum, plasma, whole blood and other fluids. 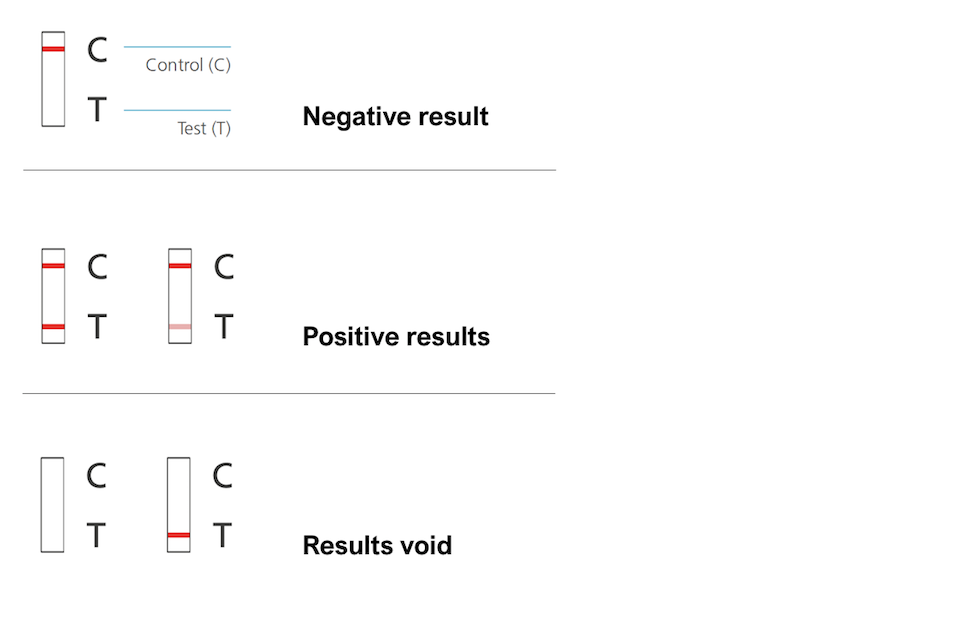
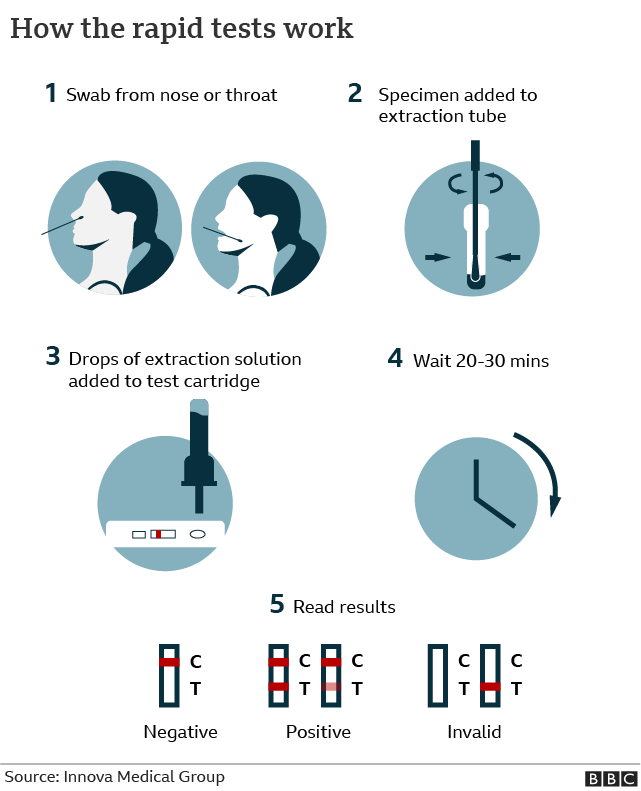
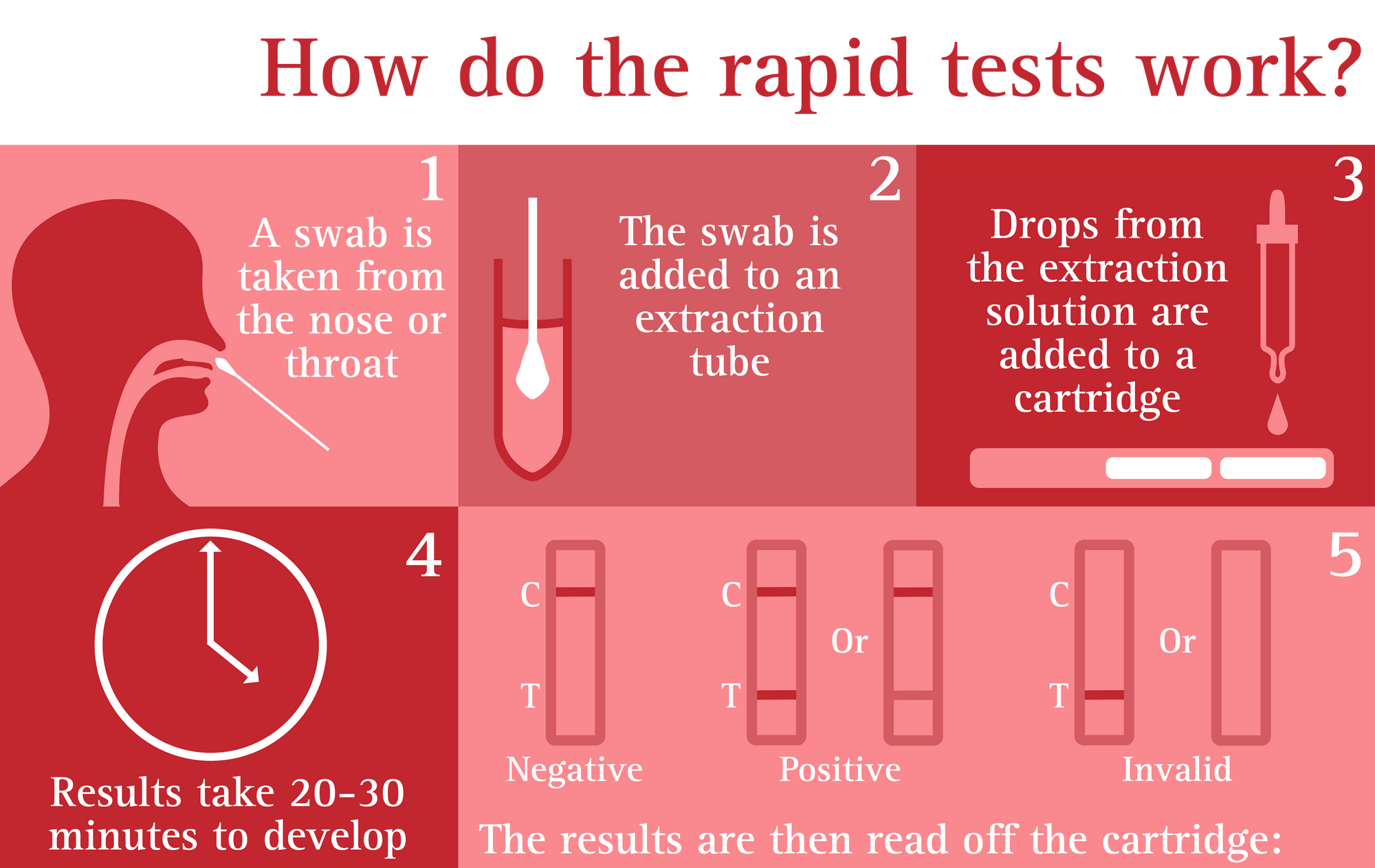
LFA-based tests are widely used in hospitals, physician's offices and clinical laboratories for the qualitative and quantitative detection of specific antigens and antibodies, as well as products of gene amplification. Low development costs and ease of production of LFAs have resulted in the expansion of its applications to multiple fields in which rapid tests are required. PHS is encouraging everyone to report their LFD test data so that an accurate report of testing that is ongoing in the population can be captured.The lateral flow assay (LFA) is a paper-based platform for the detection and quantification of analytes in complex mixtures, where the sample is placed on a test device and the results are displayed within 5–30 min. The data published on Thursday using this new measure will cover testing results up to Tuesday 11 January. 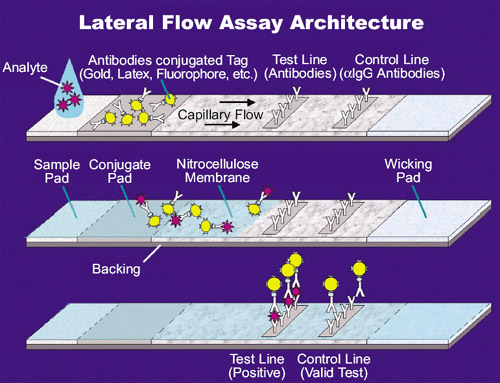
Given the increase in data to be interrogated, there will be a lag in reporting while PHS works to streamline the process. LFD positive cases that are followed by a negative PCR result within 48 hours will be denotified. From Thursday 13 January, PHS will also begin reporting on the number of people with a COVID-19 infection confirmed by either a PCR or first LFD positive test. In order to ensure that Public Health Scotland (PHS) continues to provide the most accurate information, changes have been made to the national COVID-19 case definition to reflect this revised testing strategy.Īt present, PHS reports on PCR tests only. Instead, anyone with a positive LFD should report the result online as soon as the test is done. On 5 January 2022, the Scottish Government announced that people will no longer be asked to take a polymerase chain reaction (PCR) test to confirm a positive Lateral Flow Device (LFD) result.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
